Innovation in parasitic worm vaccine research: platforms used in WORMVACS2.0
Around the world, people continue to suffer from the effects of parasitic worm infections caused by soil-transmitted hookworms and snail-borne schistosomes. Despite the devastating effects on public health – most pronounced in low- and middle-income countries – these infections are often under-researched and remain some of the world’s most devastating and persistent Neglected Tropical Diseases.
WORMVACS2.0 researchers have set out to change this. So far, no effective vaccines against parasitic worms exist, and the international consortium aims to establish an effective pipeline for their development. To do so, the researchers are exploring and developing three innovative vaccine production platforms that have not yet been applied in worm vaccine development: recombinant glycoproteins produced in plants, mRNA, and outer membrane vesicles.
Each of them offers potential benefits, and looking at them together as part of WORMVACS2.0 gives researchers a unique opportunity to advance the search for a parasitic worm vaccine.
Below, you can read more about all three platforms used in WORMVACS2.0. If you are interested in recent project milestones, make sure to check out the news page as well, or explore project publications.
Lead project partner: Wageningen University
What is this approach, and how is it used in vaccine production?
We all know plants for their important role in nutrition. But next to that, plants are also a great platform to produce recombinant proteins for vaccines. In that way, they kind of turn into a living bioreactor. So, whereas in traditional systems, proteins for vaccines are produced by growing bacteria or other cells in large reactors, we use plants for production.
Explore the process through the photos:
What are the benefits of this approach?
A big advantage of using alternative production systems to produce the vaccine is that you do not need to extract the proteins from the parasite. If we didn’t have recombinant production systems, we would need to sacrifice many animals to produce enough live parasites to extract the proteins needed for the vaccines. This makes plants a cheaper and friendlier production system.
On an evolutionary basis, plants are more similar to helminths than bacteria, and they are rather robust against changes in their genome, so you can remove or add certain things from the vaccine. Therefore, this approach can be used when the vaccine requires specific characteristics that other production platforms do not offer. Another big advantage is that the plants that we use do not produce endotoxins that can be harmful to humans, making it a very safe platform.
How is it used in WORMVACS2.0?
In WORMVACS2.0, this platform is used by producing secreted glycoproteins from Schistosoma mansoni or Necator americanus with glycans that are found in those parasites. We will create a set of vaccines containing the same protein with different glycosylation to determine the role of these glycans in immune recognition and vaccine function.
Lead project partner: Leiden University Medical Center
What is this approach, and how is it used in vaccine production?
Many of us know how effective mRNA vaccines can be from personal experience during the COVID-19 pandemic. The relatively novel mRNA platform technology was a game-changer that allowed the rapid development of a vaccine against the SARS-CoV-2 virus by BioNTech and Moderna.
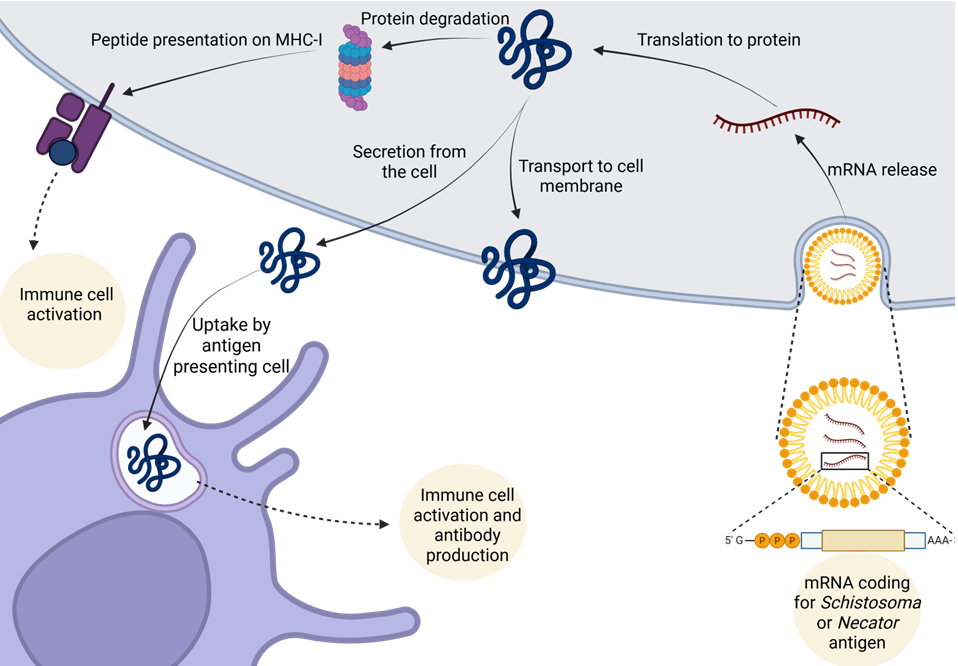
The approach is based on immunisation with lipid particle- enclosed mRNA that encodes the amino acid sequence of a specific pathogen protein, such as the viral spike protein in the case of SARS-CoV-2. The mRNA is taken up by cells in the immunised person and then translated into the pathogen protein it encodes. This antigenic protein is then presented to the immune system, which mounts a protective response consisting of antibodies and/or cellular immunity.
Lipid-nanoparticle encapsulated mRNA encoding the S. mansoni or N. americanus vaccine target is taken up by recipient cells, which translate the mRNA sequence into protein. Depending on the construct, the protein is exposed to the immune system as a secreted or membrane bound antigen. The immune system responds by generating parasite-specific antibodies and immune cells.
What are the benefits of this approach?
A major benefit of this approach is that the immunogen (the targeted pathogen protein) is produced within the immunised subject itself. This means that the protein does not have to be produced in a recombinant form, which can be difficult.
The production of mRNA is easier and quicker, and it also allows much more flexibility in creating different variations of the antigen for pre-clinical testing. It is, for instance, relatively easy to test partial sequences or include variations such as secreted or membrane-bound versions of the same proteins to optimise the efficacy of the vaccine. Depending on the pathogen and the protective mechanism, it may be relevant whether the antigen is presented on the cell surface or not.
The approach is also well-suited for combining different mRNA sequences into a single vaccine formulation to create multivalent vaccines that target multiple antigens in a single immunisation.
How is it used in WORMVACS2.0?
WORMVACS2.0 participants have teamed up with Moderna to apply their mRNA platform via the mRNA Access program (https://mrna-access.modernatx.com/). To date, there is very little information about the potential efficacy of mRNA vaccines against parasitic worm infections. The question is whether the mRNA approach for existing or new targets against schistosomes or hookworms can be effective, and how this compares to other platforms.
Prioritised vaccine targets that WORMVACS2.0 partners identified through antibody analyses in controlled human infection studies and endemic populations, as well as literature data and bioinformatics, are provided to the project by Moderna as mRNA constructs. These constructs may encode full-length proteins of S. mansoni or N. americanus, or partial sequences such as only the functional domain of a protein, and we often test both the secreted and membrane-bound versions by specifically including or deleting a transmembrane domain and signal peptide sequences.
Such non-formulated constructs are tested for in vitro expression in human or rodent cell lines to ensure proper production of the encoded protein in the recipient cell.
Subsequently, the mRNA Access program supports the production of the selected mRNA constructs in a formulated nano-lipid particle, which can then be used in pre-clinical testing in rodent models. First immunisations are carried out to ensure that antibodies are produced, indicating that a proper immune response is mounted. Then, a subsequent challenge with infectious parasite larvae is done to test the protective efficacy of the immunisation by determining a possible effect on worm development and survival.
Schistosome vaccines are tested by WORMVACS2.0 partners at Leiden University Medical Center, Aberystwyth University, Texas Tech University System.
Hookworm vaccines are tested by the Swiss Tropical & Public Health Institute.
If this pre-clinical data shows good effectivity, WORMVACS2.0 is aiming to move one S. mansoni vaccine candidate as an mRNA vaccine into a small-scale safety trial in humans.
Lead project partner: BiOMViS
What is this approach, and how is it used in vaccine design?
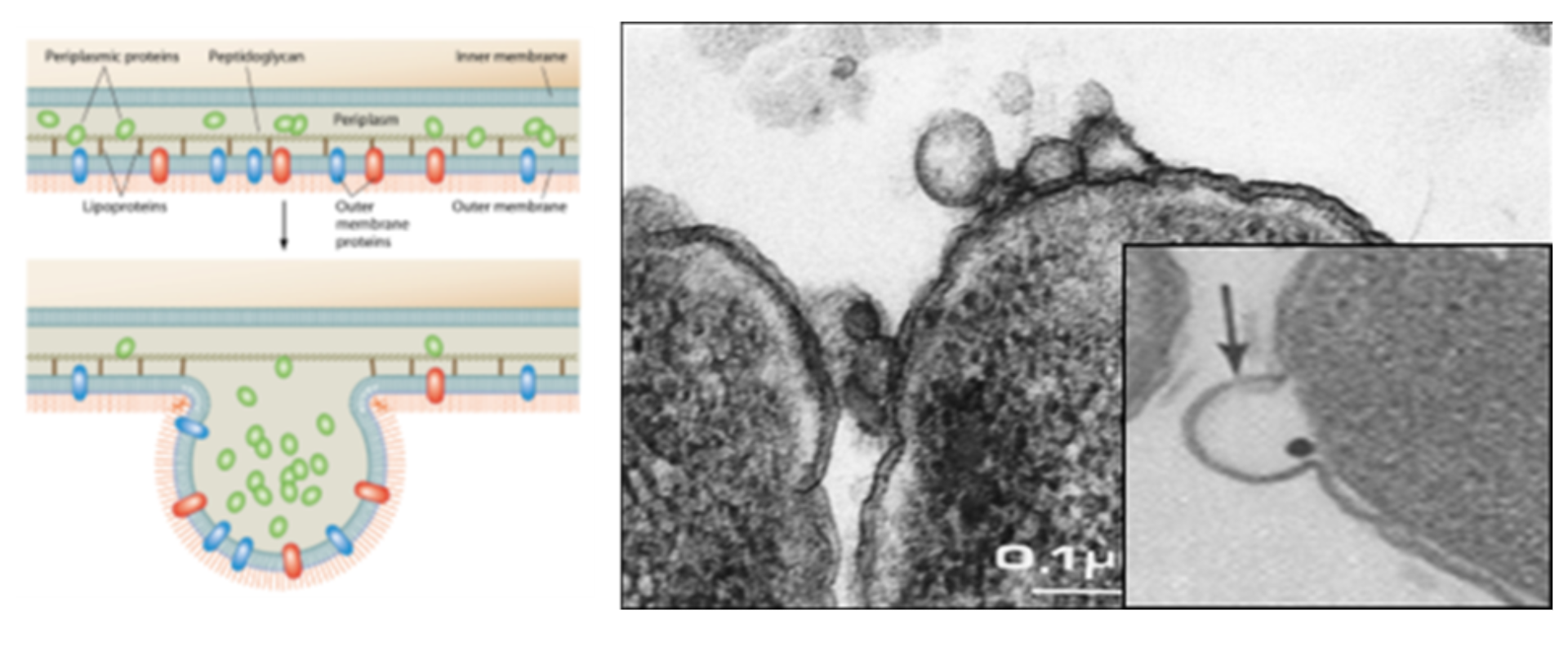
Outer Membrane Vesicles (OMVs) are emerging as a promising platform for next-generation vaccines thanks to their unique properties. These bacterial structures act as natural adjuvants, carrying molecules (MAMPs) that strongly stimulate the immune system, particularly Th1-type responses.
A schematic reconstruction of the mechanism by which bacterial outer membrane vesicles are released and an electron microscope image of a Gram-negative bacterium releasing OMVs (indicated by the arrow in the enlarged detail panel).
Through genetic engineering, OMVs can also be designed to display specific antigens on their surface, effectively combining immune activation and antigen delivery in a single, ready-to-use system.
However, some challenges remain. Native bacterial proteins within OMVs can dilute the target antigens, reducing their impact, and methods for loading foreign proteins still require optimization to ensure efficiency and proper antigen structure.
Finally, key components such as lipopolysaccharide (LPS) must be carefully controlled to balance strong immune stimulation with acceptable safety.
In short, OMVs represent a highly promising vaccine platform, but further fine-tuning is needed to unlock their full potential.
What is the novelty of the BiOMViS OMV-based vaccine platform?
Over the last several years, BiOMViS has been dedicating substantial efforts to establishing an Outer Membrane Vesicles (OMV)-based vaccine platform that could overcome the limitations of the technology and become an attractive option for designing highly effective, safe, and inexpensive vaccines.
In particular, the OMV-based platform developed by BiOMViS is based on two key technological pillars:
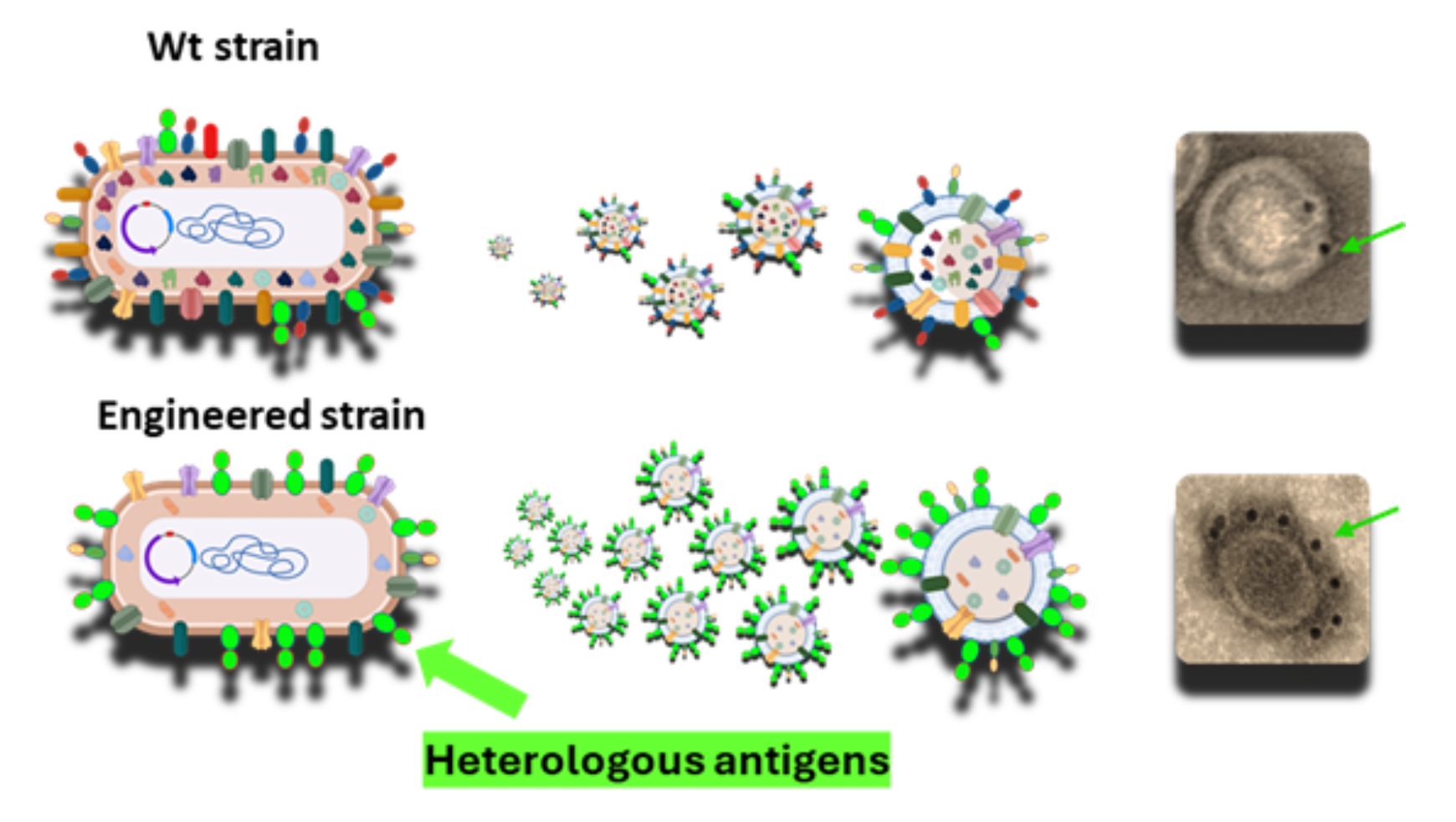
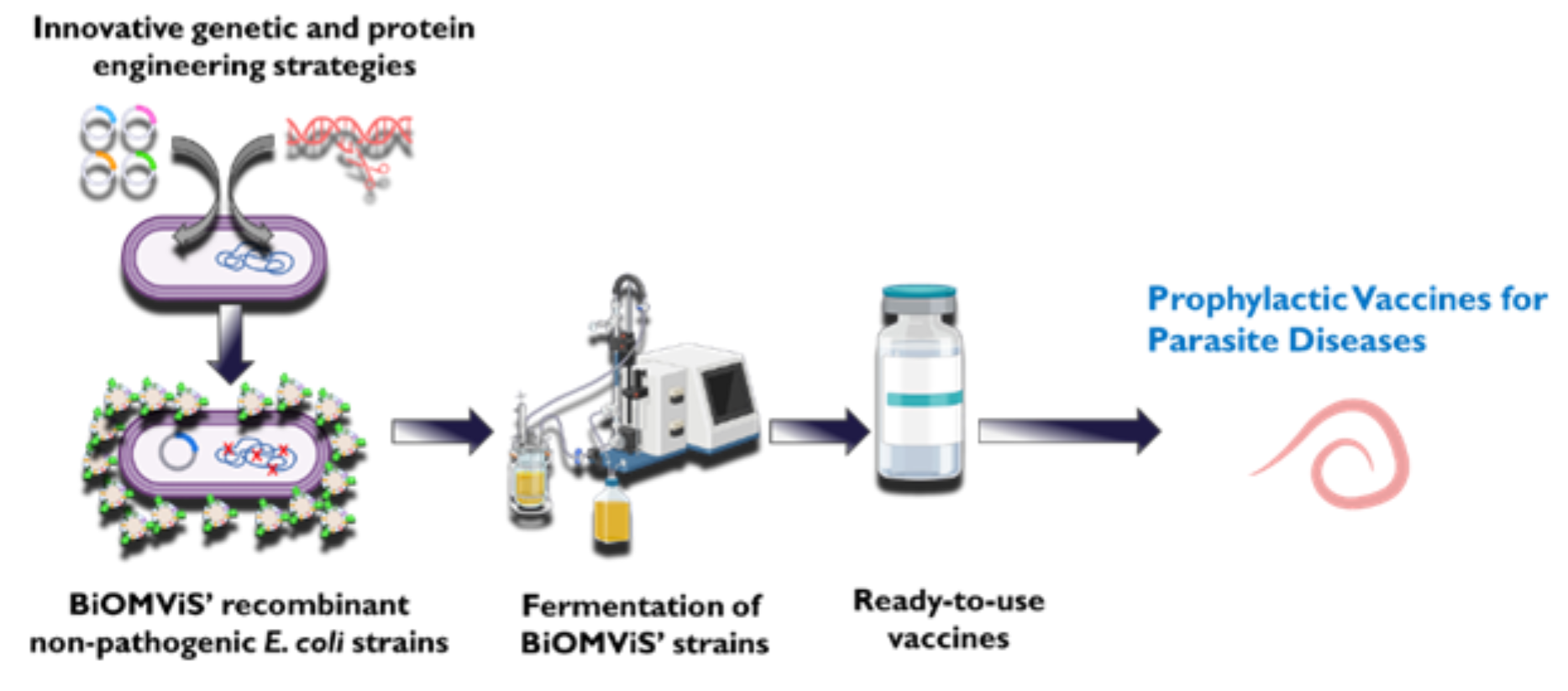
1. OMV producing strains: using synthetic biology, BiOMViS has created a set of proprietary non-pathogenic E. coli strains, which release abundant quantities of vesicles deprived of a large number of endogenous proteins. The absence of these proteins enables high levels of expression and immunogenicity of heterologous antigens.
Moreover, the strains also carry mutations in genes involved in the lipopolysaccharide (LPS) biosynthesis, so that the released OMVs contain detoxified versions of the Lipid A. This detoxification has resulted in an excellent in vitro and in vivo safety profile to the OMV-based vaccines, while preserving high adjuvanticity.
2. OMV engineering: BiOMViS has also set up genetic engineering strategies for efficiently foreign proteins (antigens) into OMVs. The gene of interest is inserted into a battery of proprietary expression plasmids which are subsequently used to transform BiOMViS OMV-producing strains. The most efficient expression system is then selected by monitoring the accumulation of the protein in the vesicular compartment.
BiOMViS-engineered E. coli strains produce increased quantities of OMVs with enhanced expression of the target antigen compared to native strains. Electron microscopy images highlight greater antigen accumulation (green arrow) in BiOMViS-derived OMVs.
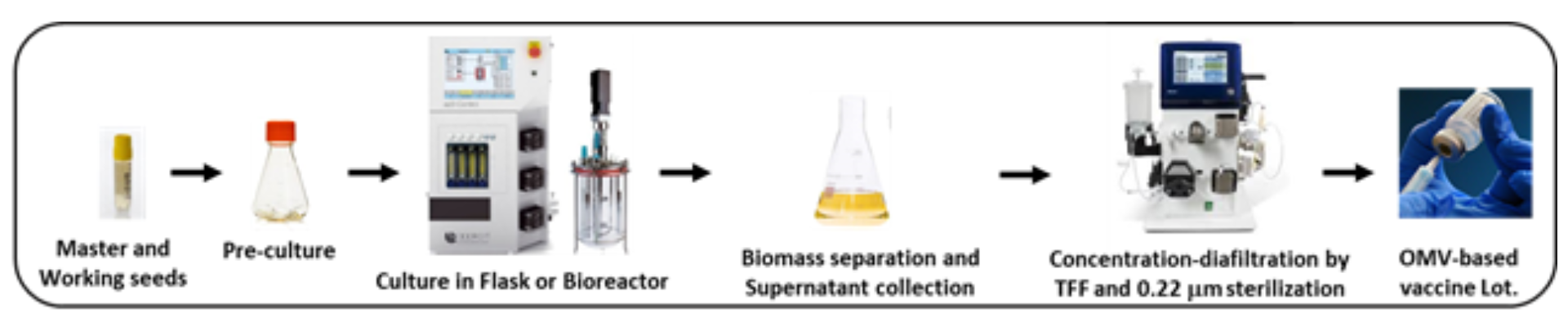
3. Efficient and robust OMVs production process: BiOMViS has also set up a process for the production and purification of the OMVs from the culture supernatant. The OMV-overproducing strains are grown in liquid cultures, and the vesicles are collected from the culture supernatant using tangential flow filtration (TFF). Yields higher than 100 mg of vesicles per litre of culture are routinely obtained.
BiOMViS process flow for recombinant OMV-based vaccine production, highlighting key advantages of simplicity, scalability, and high yield of vaccine doses.
How is the BiOMViS platform used in WORMVACS2.0?
The BiOMViS OMV-based platform is being applied within the WORMVACS2.0 project to develop innovative vaccines against human-infecting parasitic helminths, with a focus on schistosomes and hookworms, some of the world’s most persistent and devastating Neglected Tropical Diseases (NTDs).
In previous studies, it was shown that schistosome and hookworm infections induce a certain level of natural immunity that is mediated by immune responses against a few specific antigens. Moreover, it was shown that vaccines formulated with such antigens (“model antigens”) can elicit partial protection in several animal models of helminth challenge.
Based on these data, one of the objectives of the WORMVACS2.0 project is to test whether BiOMViS OMVs decorated with model antigens and novel antigens discovered during the project can be used to develop novel parasitic vaccines. More specifically, antigens will be expressed in BiOMViS OMVs and after purification, the engineered OMVs will be tested for their capacity to induce protective immunity in mouse and guinea pig models of helminth infections.
Unique features of the BiOMViS platform—engineered OMV-producing strains and methods for incorporating helminth-specific antigens—together with a streamlined production process, enable rapid vaccine development against parasitic infections, supporting a key WORMVACS2. 0 project objective.
The availability of an effective OMV-based vaccine would offer a number of advantages. Considering the simplicity of the production process, the large number of doses available at very low cost, and the stability of the formulations – cold chain not required - these vaccines are particularly well suited for low-income countries, where the highest annual mortality from these parasitic infections is recorded.